
Why is a Diamond of Type IIb blue to greyish blue?

by J.-P. Chalain first published in Facette 25 (February 2019)
When a chemically pure diamond is only made of carbon atoms, it iss classified as type IIa, and when crystal defaults are absent, it is colourless.
In a type IIb diamond some boron atoms substitute with diamond’s carbon atoms and a very weak concentration of boron – in the order of a few ppb (a few boron atoms per billion carbon atoms) is sufficient to create a bluish grey, greyish blue to blue colour. This is the case of some very famous diamonds such as the Hope diamond or the Wittelsbach- Graff diamond.
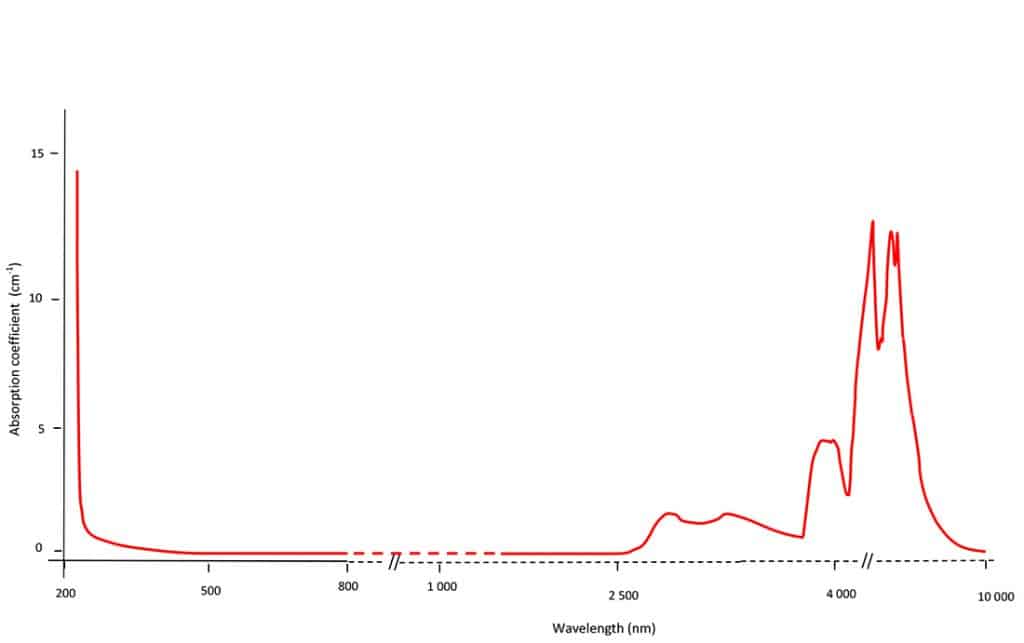
Looking at the absorption spectrum of a type IIa diamond between 10’000 nm to 200 nm (Figure 1), one notes that this diamond does not absorb any light between 2’500 nm and 230 nm. At these wavelengths the type IIa diamond is perfectly transparent and this includes the visible range wavelengths, between 800 nm and 400 nm that human eyes are sensitive to- hence its perfect colourlessness (D colour grade). Interestingly, in the mid-infrared (between 2’000 nm and 7’000 nm) the pure type IIa diamond absorbs a lot of light; these absorptions are due to chemical bonding of carbon atoms between themselves, hence the name of this region: diamond’s intrinsic absorption region.
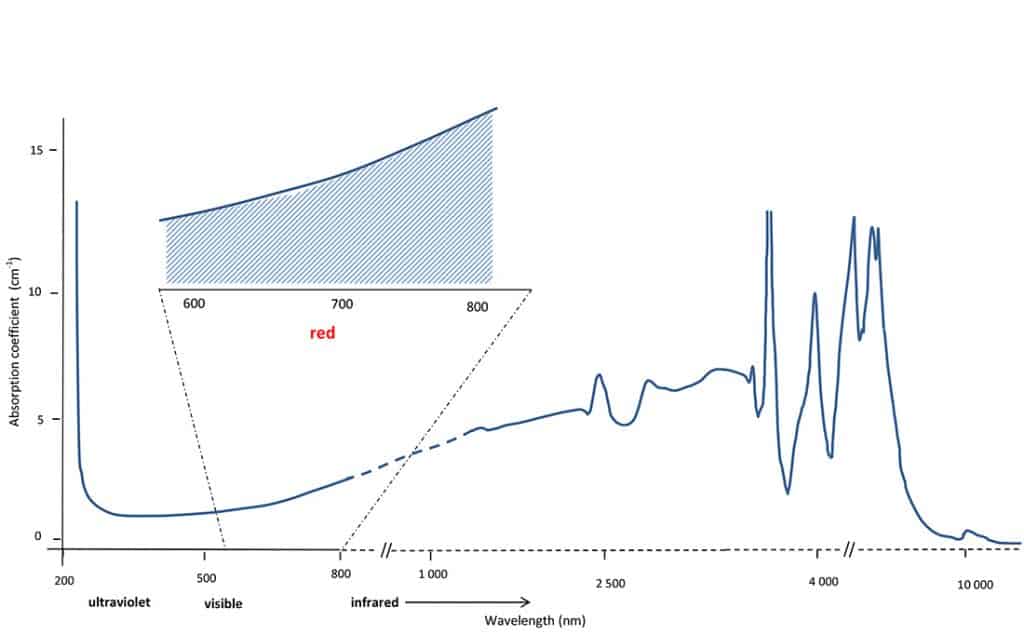
Now, looking at the absorption spectrum of the greyish blue type IIb diamond (Figure 2), one notes that this last absorption region is greatly different than that of the chemically pure type IIa diamond. The difference is due to the presence of boron atoms which greatly modifies the diamond’s intrinsic absorption region. For instance, the type IIa diamond at 2’500 nm does not absorb any light (the absorption coefficient at this wavelength is nil; and this is also the case of most other diamonds of type I) but at the same wavelength, the diamond of type IIb absorbs a lot; up to an absorption coefficient of 5. Looking further at smaller wavelengths we see that this type IIb absorption regularly decreases towards the visible light region and still absorbs in the red part (800 to 600 nm) of the visible range.
The diamond’s absorptions in the visible range give a direct explanation of the diamond’s colour. One should remember that:
(1) a human being’s eyesight is more sensitive to yellow
(2) any transparent material illuminated with white light and absorbing a specific colour will appear in the complementary colour
For instance, a diamond absorbing wavelengths in the blue range will be seen yellow; a diamond absorbing wavelengths in the red range will be seen blue and a diamond absorbing all wavelengths of the visible range will be seen grey to black.
Having a last look at the type IIb absorption spectrum, we now understand that this type IIb diamond is greyish blue for two reasons: 1) the tail of absorptions due to the presence of boron absorbs in the whole visible range (between 2 and 3 in absorption coefficient), hence its greyish hue and 2) absorbs more in the red than in other ranges of the visible spectrum, hence its bluish hue. The combination of the two (see the striped zone in the inset of Figure 2) results in the typical greyish blue colour of such a type IIb diamond.
Want to learn more about diamonds?

Sign up for our free online course: Introduction to diamonds


